Electrolysis
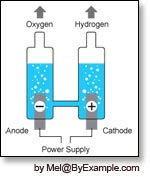
This is an illustration of the chemical process of electrolysis in which electricity is used to split water into separate molecules of hydrogen and oxygen.
-
Electrolysis Diagram

- Electrolysis is a chemical process that can be used to split water molecules into hydrogen (H) and oxygen (O). During electrolysis electricity is channeled into a chamber of water. The water acts as a conductor allowing electricity to pass between two electrodes, which divides the H2O into individual hydrogen and oxygen molecules. Once freed, oxygen bubbles collect on the anode and hydrogen on the cathode.
-
Electrolysis Diagram (preview)
- Preview version of the electrolysis diagram.